kpk1220
the Technology Interface / Winter99
Applications & Developments in the Cryogenic Processing of Materials
by
Kristoffer P. Kollmer
colonel6@earthlink.net
Mechanical Engineering Technology
Central Washington University
Abstract:
The technique of cryogenic processing is an inexpensive method that improves the physical and mechanical properties of materials such as metals, plastics and composites. Published research about the cryogenic processing of materials is nearly unavailable. However, this is a field of engineering that is growing rapidly and is used by many manufacturers. I plan to construct a facility that will treat materials in order to research both the process itself and the results of the processing. This may help to create standards for both processing and testing that are currently unavailable.
- Project Description
Objectives:
1. To design and build a small scale working facility for the cryogenic processing of materials
2. To treat standard test specimens and perform a physical and metallurgical analysis, comparing this to identical specimens that are untreated and specimens that are commercially treated. Testing will include:
n
Static Tensile Test
n
Rockwell Hardness Test
n
Charpy Impact Test
n
R.R. Moore Fatigue Test
n
Microscopic Analysis of Grain Structure Changes in Treated Materials
3. To enhance the knowledge of cryogenic processing through the development of specific testing methods and standards adapted from existing standardized testing methods. A comprehensive report on the results of this project’s design, construction and material analysis will be submitted to active engineering organizations for publication.

Figure 1: Tool Steel Punches:
These two punches were used together on identical materials. The punch on the left had been cold tempered before use and the punch on the right was used without any modification. The punch on the right shows much more wear and is chipped along the edge, while the cold tempered punch is still usable.
- Introduction
The use of cryogenic fluids to increase the physical and mechanical properties of materials is a technology that has experienced rapid development within the last few years. This technique is called cryogenic processing or cryogenic tempering. It involves slowly cooling a material, such as steel or plastic, from room temperature to around -300ºF and then soaking or holding the material at this temperature for a specific time. The material is then brought back to room temperature. This process refines and stabilizes the crystal lattice structure (see figure 2 below) and distributes carbon particles evenly throughout the material creating a stronger, more durable material. This permanent change relieves nearly all of the residual stresses introduced by casting, forging, machining or heat treatment. Quantified studies on the results of this treatment are nearly nonexistent for this process. Because of this, I am designing and building a facility to treat materials and to perform a quantitative analysis of the results. This project is a culmination of everything I have learned during my studies in mechanical engineering technology and has implications for both advanced research and future employment. I intend to process standard test specimens and test them for hardness, tensile strength, fatigue strength and impact strength. I also plan to do a metallurgical and image analysis on treated and untreated materials comparing grain sizes, grain orientation, and grain boundary reduction. In order to validate my tests, I plan to analyze identical specimens that will be commercially cryotempered so I can compare both the processes and results of my apparatus versus a commercial apparatus. This analysis is intended to increase the overall knowledge of cryogenic treatment and cryogenic engineering, which is a field that lacks both standardization and published research. Also, since this facility will remain at Central Washington University, it can be integrated into future experiments where students can learn how the process works and its applications in engineering.
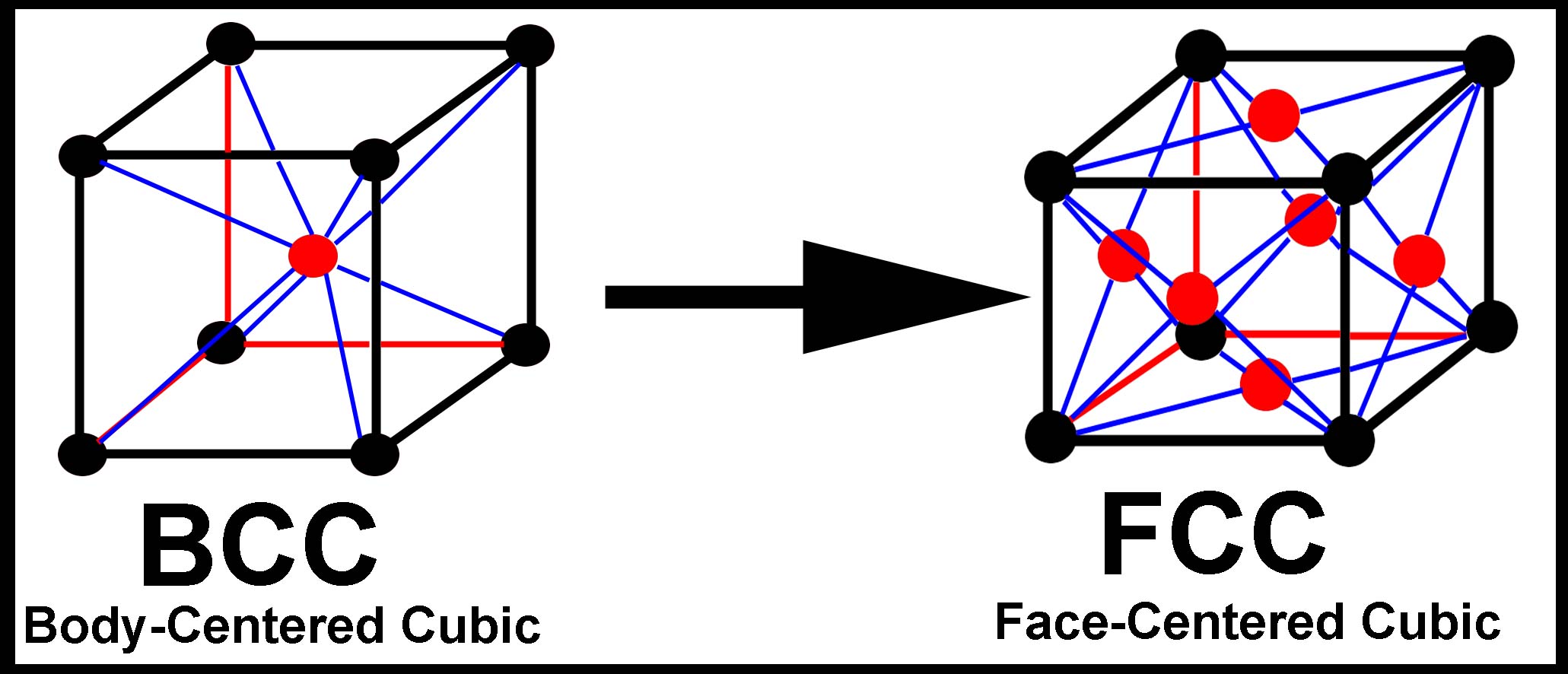
Figure 2
Crystal Lattice Structures & Cryogenic tempering
During the Cryogenic Tempering Process a material such as steel goes through a phase change that transforms the crystal lattice structure from body-centered cubic to face-centered cubic. The face-centered cubic structure has less space available for interstitial defects and results in a stronger, more durable material.
Cryogenic Treatment Facility Design & Construction
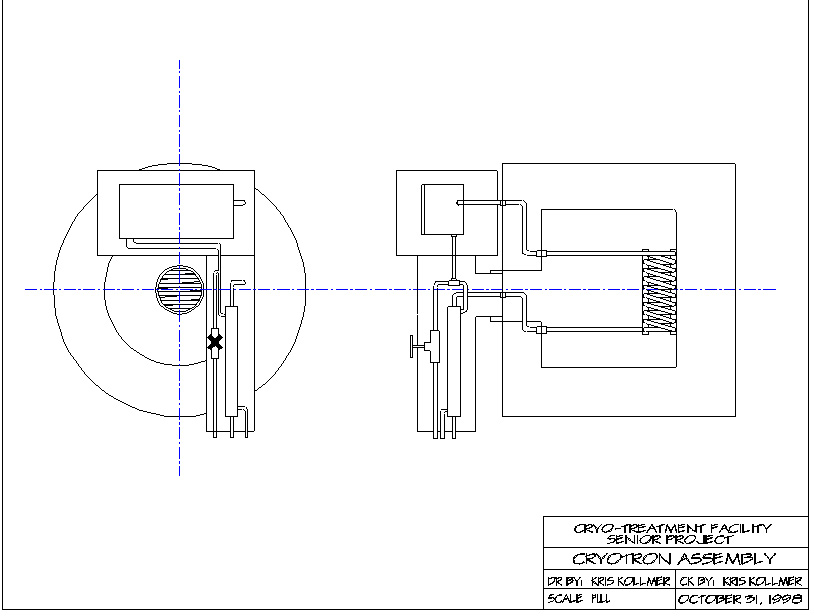
Figure 3
Cryogenic Treatment Facility Assembly Drawing
The cryogenic treatment facility built in this project weighs about 200 pounds and cost around $600 to build.
The first stage of construction was to make the main heat exchanger. The main heat exchanger consisted of 25 feet of 3/8 type L copper tubing. Thirteen coils fit the contour of the 304 stainless steel shell. The coils were kept in place with aluminum bars with holes drilled for the tubes. The aluminum bars were bound with solid copper wire. The next step was completing the dewar construction. The outer shell of the dewar was a 55 gallon steel drum which was cut to 22 inches. This allowed for the correct amount of insulation. One of the more difficult parts of the dewar was to incorporate a vacuum in the outer shell to eliminate moisture and condensation contamination of the insulation. The inner shell could not be sealed because as the liquid nitrogen evaporated, the pressure would cause the container to explode. The filling tube and fittings were first welded to the lid of the inner vessel. Next, the lid of the outer shell was welded to the filling tube. Fittings were welded to the lid of the outer shell and tubing was connected to the fittings on the inner and outer shell. The final stage of the dewar construction was to weld the outer shell and fill the inside with insulation. After the dewar was completed, the cooling box was constructed. The inner box was made of 304 Stainless Steel (18% Chromium, 8% Nickel). The box was cut from 1/4-inch sheet stock and Gas Tungsten Arc Welded together. The top of the box was milled flat on a vertical milling machine. The lid of the box was cut from 1/2-inch sheet stock and the edge was milled to produce a lip to fit onto the box. Holes were drilled and tapped to accommodate the tubing, fittings and a J-type (Iron-Constantin) thermocouple. The outer box was a steel toolbox that is the correct size to allow for insulation. The box is insulated with a combination of perlite, polystyrene and expanded foam. Holes were drilled in the box to connect the tubing coming out of the dewar and going out to the input heat exchanger. The input heat exchanger (counter-flow) was constructed from one and a half feet of one-inch copper tubing with 3/8 inch copper tubing running through the interior of the large tube. Cold exhaust gas from the cooling box comes in at one end of the tube and exits at the other end. The amount of gas entering the heat exchanger is controlled by a valve connected to a T-joint that splits the flow to the exhaust and heat exchanger. This increases the efficiency of the machine. Helium gas was used because of its high thermal conductivity and low freezing point. The input heat exchanger was encased in a six-inch schedule 80 PVC pipe and insulated with perlite and expanded foam.
IV. Materials & Methods
Physical Testing:
All testing for this project was done in the Hogue Technology building at Central Washington University. Three groups of test specimens were used. The first group was untreated, the second was commercially cold treated by OneCryo in Puyallup, Washington and the third group was cold treated using the facility designed and built in this project. All of the specimens were standard test specimens ordered from Instructional Test Equipment in Auburn, California. They were made from cold rolled 4140 steel. Impact testing was conducted on a Tinius-Olsen Charpy/Izod test machine according to ASTM E-23 standards. The machine was calibrated to determine any offset just before testing. Tensile testing was done on a Tinius-Olsen tensile/compression machine according to ASTM E-8. A Wilson Rockwell model hardness tester was used with a C-brale penetrator to determine the hardness of the material. The hardness tester was calibrated with a C-block.
Image Analysis:
Small pieces were cut off the specimens using an abrasive cutoff wheel. These pieces were mounted in phenolic preforms (Figure 4) and sanded descending from 120 to 600 grit sandpaper. They were then polished using alumina powder ranging from 15 to 0.3 microns. The specimens were cleaned using an ultrasonic bath just prior to etching. Etching was done using a 2% Nital solution for approximately five minutes. The specimens were viewed using a Leica LMDM model microscope with a Pixera CCD that transferred the images to a computer. A picture was also taken of a 1/100 inch ruler and superimposed on the specimen images to verify magnification and grain sizes.

Figure 4
Phenolic preforms holding test specimens for image analysis.
Cold Tempering Methods:
Determining the schedule for cold treatment depended mainly on three variables. These were:
- Type of Material
- Cross Sectional Area of Material
- Application for Material
4140 steel is a Chromium-Molybdenum alloy containing 0.41% carbon, 0.85% Manganese, 0.02% Phosphorous, 0.03% Sulfur, 0.20% Silicon, 0.12% Nickel, 1.01% Chromium and 0.24% Molybdenum. Molybdenum is an element that can increase the hardenability of the steel. There is no critical temperature published in the cryogenic range for this material, however, it is safe to assume that its transformation temperature is above that of liquid nitrogen. The cooling and heating ramp rate depends mostly on the cross sectional area. The test specimen cross-sectional area is small, so a temperature change of two degrees per minute is sufficient. For this application, a through hardness is required. This material has a thermal conductivity of 63 W/m-K at 74 K. This increases with temperature and is about 25% greater than 304 Stainless Steel (Barron pg. 21). This factor decreases the amount of soaking time. Where the soaking time for stainless steel is about 24 hours, the soaking time for 4140 is six to ten hours. With cooling and heating taking about three hours, one bottle of helium allows for nine hours of soaking time at 30 psig, although this pressure does not remain constant during the tempering process. Cooling the material at two degrees per minute will require between 15 and 60 psi, depending on how much heat is exchanged in the input heat exchanger. Soaking will require shorter bursts of gas, because the stainless steel box does not conduct heat well at low temperatures. The insulation outside the box has a thermal conductivity of between 0.02 and 0.08 W/m-K (because of combined insulation). The tempering schedule used for the 4140 test specimens is shown below in figure 5.

Figure 5
Cold tempering schedule for 4140 CR test specimens treated in this project.
V. Data
Physical Testing:
Tensile:
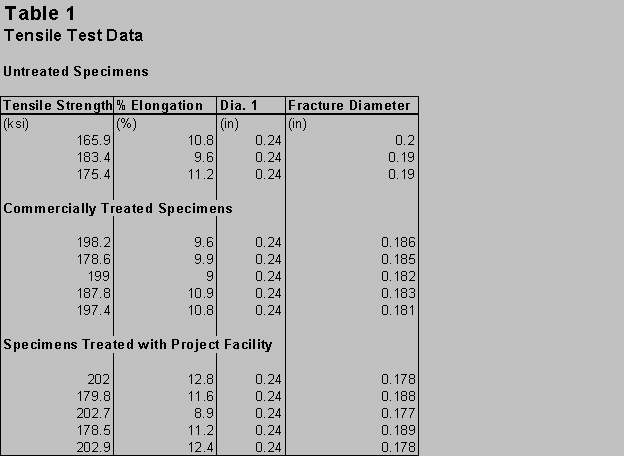
Tensile strength increases ranged between 7% and 16% for cold tempered specimens. The diameters of the cold treated specimens at the fracture point were smaller than in the untreated specimens. The fractures were a "cup and cone" typical of ductile materials. Cold treated specimens were able to withstand more elastic deformation than untreated specimens. Table 1 shows tensile data for the tensile specimens.
Charpy Impact Data:
There was almost no change in impact strength between treated and untreated specimens. The difference was shown in the fractures of the specimens. Viewed under a stereoscope, the fractures in the cold tempered specimens were rougher and had less stress hardening than the untreated specimens which suggests an increase in toughness. Table 2 shows the charpy impact data.

Rockwell C Hardness Data:
There was no significant change in the hardness of the material. However, the hardness was much more consistent in the treated specimens. In the untreated specimens the hardness varied in different places even when the St. Venant’s Principle for hardness was used. Rockwell C hardness data is shown below in table 3.
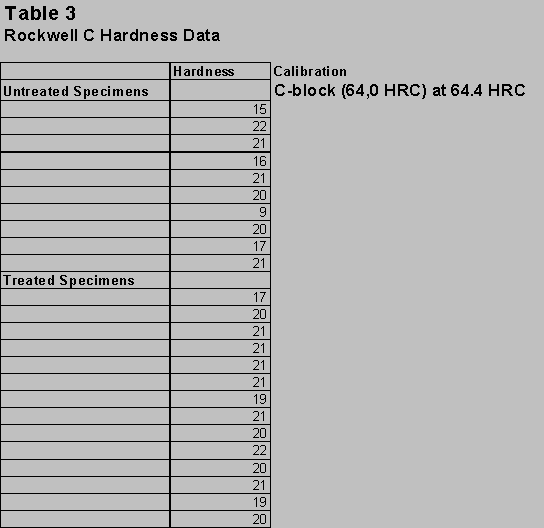
Image Analysis Data:
Grain structures were viewed on the Leica LMDM microscope at 500 X magnification. The objectives of grain structure analysis of cold treated specimens versus untreated specimens were:
- Reduction in grain size of treated specimens
- Uniform distribution of carbon particles
- Eliminate or diminish grain boundaries
Figure 6 shows the grain structure of an untreated specimen. This was cross-referenced with pictures of the same material located in the ASM Handbook. The structures were identical. However, the grain structures of the commercially treated specimen (figure 7) and the specimen treated the facility designed in this project (figure 8) could not be cross-referenced with the ASM Handbook because there were no pictures of cold treated materials in this book. The closest match to the cold treated specimens was a 4140 steel bar, austenitized at 1550 F, oil quenched to 150 F, and tempered 2 h. at 1150 F (figure 9). Grain size reduction in cold treated specimens ranged from 50% to 75% with the average grain size of untreated specimens at 7 microns and the grain size of treated specimens at 2 to 4 microns. The carbon is much more distributed in the treated specimens and grain boundaries have almost disappeared.

Figure 6:
Grain structure of untreated specimen

Figure 7:
Grain structure of specimen commercially treated at OneCryo in Puyallup, WA

Figure 8:
Specimen treated with cryotreatment facility designed and built for this project.
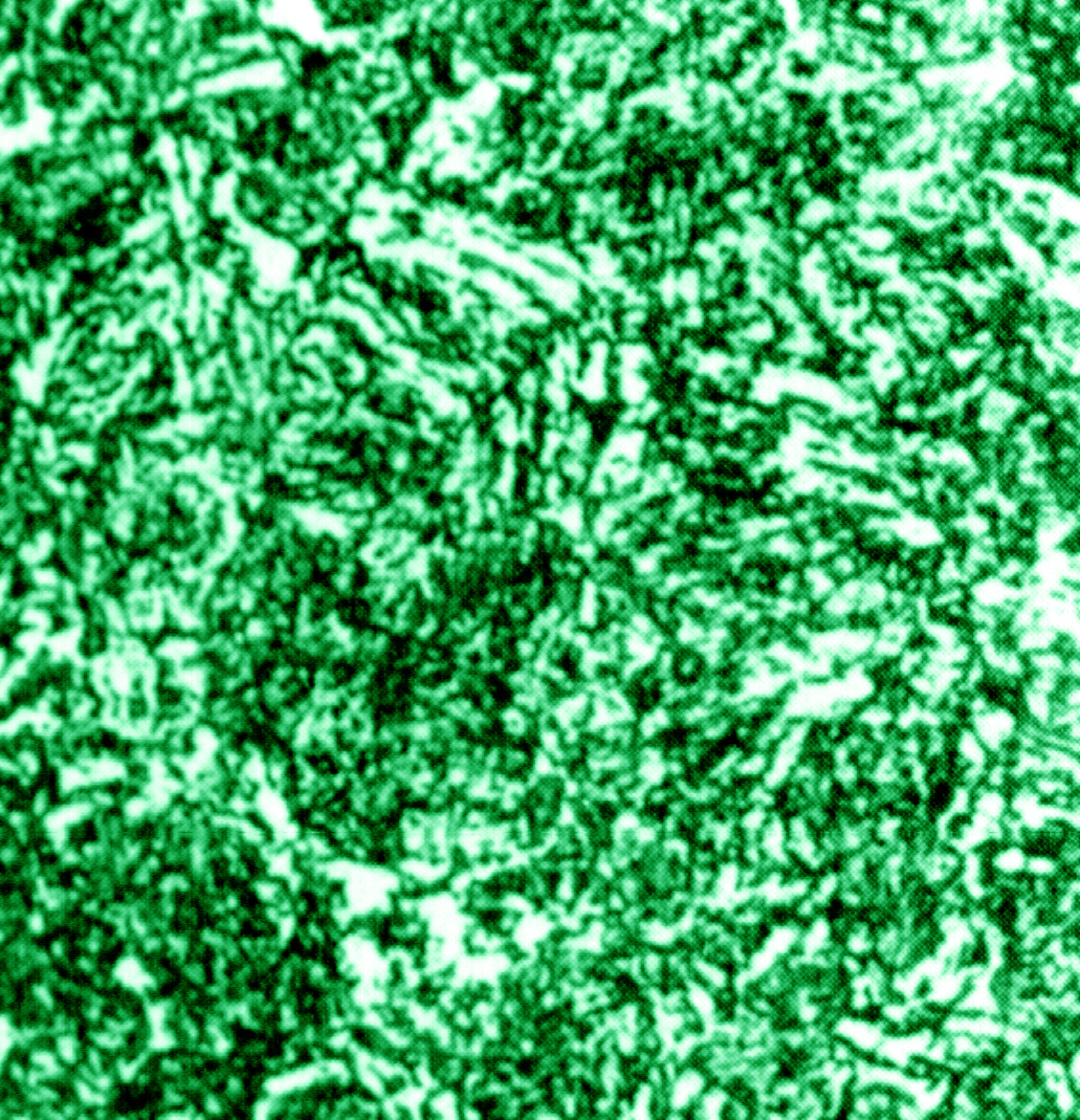
Figure 9:
4140 steel bar, austenitized at 1550 F, oil quenched to 150 F, and tempered 2 h. at 1150 F. Structure is a martensite-ferrite-carbide aggregate. 2% nital. 750X. (ASM Handbook, Vol. 9 pg. 191)
Cold Treatment Data:
The CryoTron cold treatment facility designed in this project treated the test specimens using a dry process. A dry process (in cold treatment terms) is where the treated material is not immersed in liquid. The amount of material being treated was small, so the schedule for treatment was a total of fifteen hours. The cooling ramp was set at two degrees per minute, which made up a total of three hours. The actual cooling time was about three degrees per minute. The lowest temperature reached by the machine was -286º F. +/-10ºF. This was sufficient to temper the specimens because only -270ºF was needed. The machine held this temperature for about two minutes without needing more cold helium. After about five hours at this temperature, the helium was left at 60 psi. The machine was then left overnight until the helium ran out and the specimens cooled on their own. After 15 hours the specimens were at 15ºF. The cooling box was then opened and the specimens were warmed by air to room temperature. Figures 10 and 11 show the CryoTron Cold Treatment Facility running with helium gas.
 |
 |
| Figure 10 | Figure 11 |
The CryoTron PX-1000 Cold Treatment Facility
VI. Conclusions & Recommendations
1. Cryotempering Facility Design and Construction:
The cryogenic facility designed and built in this project was capable of cold treating specimens that are up to 8"X3"X3". The cold treatment performed was thorough, treating through the whole specimen. The quality of treatment was nearly identical to commercially available treatments. The only problems with the cold treatment facility were with automation. For each different type and size of specimen, heat transfer calculations must be performed to determine:
- Cooling ramp time
- Soaking time
- Thermal conductivity of material at cryogenic temperatures
Another way to improve the design of the facility would be to add stiffeners to the outer shell of the dewar so more vacuum could be held, improving insulation. Continuing research on this project should include more microscopic analysis at different stages of the treatment. This would show exactly how the grain size decreases and carbon is distributed. This could be done by either adding a cold stage to the existing microscope or removing the specimen and quickly taking a picture of the grain structure.
2. Physical Testing
Tensile test results yielded a 7% to 16% increase in tensile strength. Although this was not a substantial increase, different materials may demonstrate larger increases. Continuing research should include testing different treated materials to determine specific improvements to each material. Cold tempering did not increase the hardness of the material. Comparing the untreated material to treated materials, the hardness was much more consistent on the treated specimens. This suggests that when the grain structure is more refined, the material is more structurally stable and residual stresses and weak points in the material have been decreased. Cold tempering also did not increase the impact strength of the material. However, there was a considerable decrease in the stress hardening area at the fracture of the specimen. This was consistent with the increased wear characteristics of cold treated materials. This may change with different materials. R.R. Moore fatigue testing was not conducted in this experiment because the testing machine was not in operating condition. This is disappointing and any further research must include this test. Fatigue specimens were treated in this experiment and are available for testing.
Microstructure analysis:
As shown previously in figures 17-20, the grain structure changes in cold treated materials were very significant. Results of microanalysis were:
- 50% to 75% decrease in grain size in treated specimens
- Much more uniform distribution of carbon
- Alleviation of grain boundaries
Grain boundaries are where stresses and defects are most likely to be. Interstitial defects can cause a material to fail prematurely, so cold treatment can decrease the likelihood of premature failure and relieve much of the residual stresses. This property is probably the biggest advantage of the cold treatment process with applications in gears, shafts, turbine blades and other critical machinery parts.
3. General Conclusions and Recommendations:
This project has definitely enhanced the knowledge of cryogenic engineering of the author and everyone else involved in the project. The largest concern is that research continues on this project to use and improve existing testing methods outlined in this research. In order to develop standards for this process, it is required to treat and test many different materials, documenting improvements much like was done for the 4140 steel.
VII. Acknowledgements
The author acknowledges and greatly appreciates the financial support offered by the Central Washington University Undergraduate Research Committee. The author would also like to acknowledge the following people for their help and support during this project:
Dr. Walter R. Kaminski, Head of Mechanical Engineering Technology program, Central Washington University, for his help and advice on the design of this project and for his continued support and encouragement throughout the project.
Dr. Craig H. Johnson, Student Advisor, Mechanical Engineering Technology, Central Washington University, for his help with materials engineering and testing methods used in this project.
Brad Campbell, Master Machinist, Central Washington University, for his help with the construction of the cold treatment facility.
Mike Carlman, Project Engineer, OneCryo, Inc., Puyallup, WA, for treating materials and encouraging research in this field.
The Industrial Engineering Technology Department, Central Washington University, for their support during this project.

VIII. References
1. Barron, Randall, Cryogenic Systems. (Oxford University Press) 1985.
2. Mott, Robert, Machine Elements in Mechanical Design. (MacMillan) 1992.
3. Holman, Jack, Heat Transfer, 7th ed. (McGraw-Hill) 1990.
4. American Society of Metals, ASM Handbook Vol. 9 (ASM International) 1996.
5. ASTM E-4, E-6, E-8, E-23. (American Society of Testing & Materials) 1978
6. Husar, John, "Cryotreatment: Panacea or Black Magic?" Cold Facts (Cryogenic Society of America) 1997.
7. Giauque, W.R. and Clayton, John, Journal of the American Chemical Society, Vol. 55 (American Chemical Society) 1933.
8. Millar, R.W. and Sullivan, J.D. Bureau of Mines Technical Paper no. 424, (Bureau of Mines) 1928.
9. Hoge, H.J. Journal of Research for the National Bureau of Standards, Vol. 44 (National Bureau of Standards) 1950.














